-
 PollenCountTODAY'S POLLEN COUNTview thepollen and moldreport
PollenCountTODAY'S POLLEN COUNTview thepollen and moldreport -
 ConvenientlyLocatedFIND YOUR CLINICCALL FOR AN APPOINTMENTOKC |Edmond | Norman |Yukon
ConvenientlyLocatedFIND YOUR CLINICCALL FOR AN APPOINTMENTOKC |Edmond | Norman |Yukon -
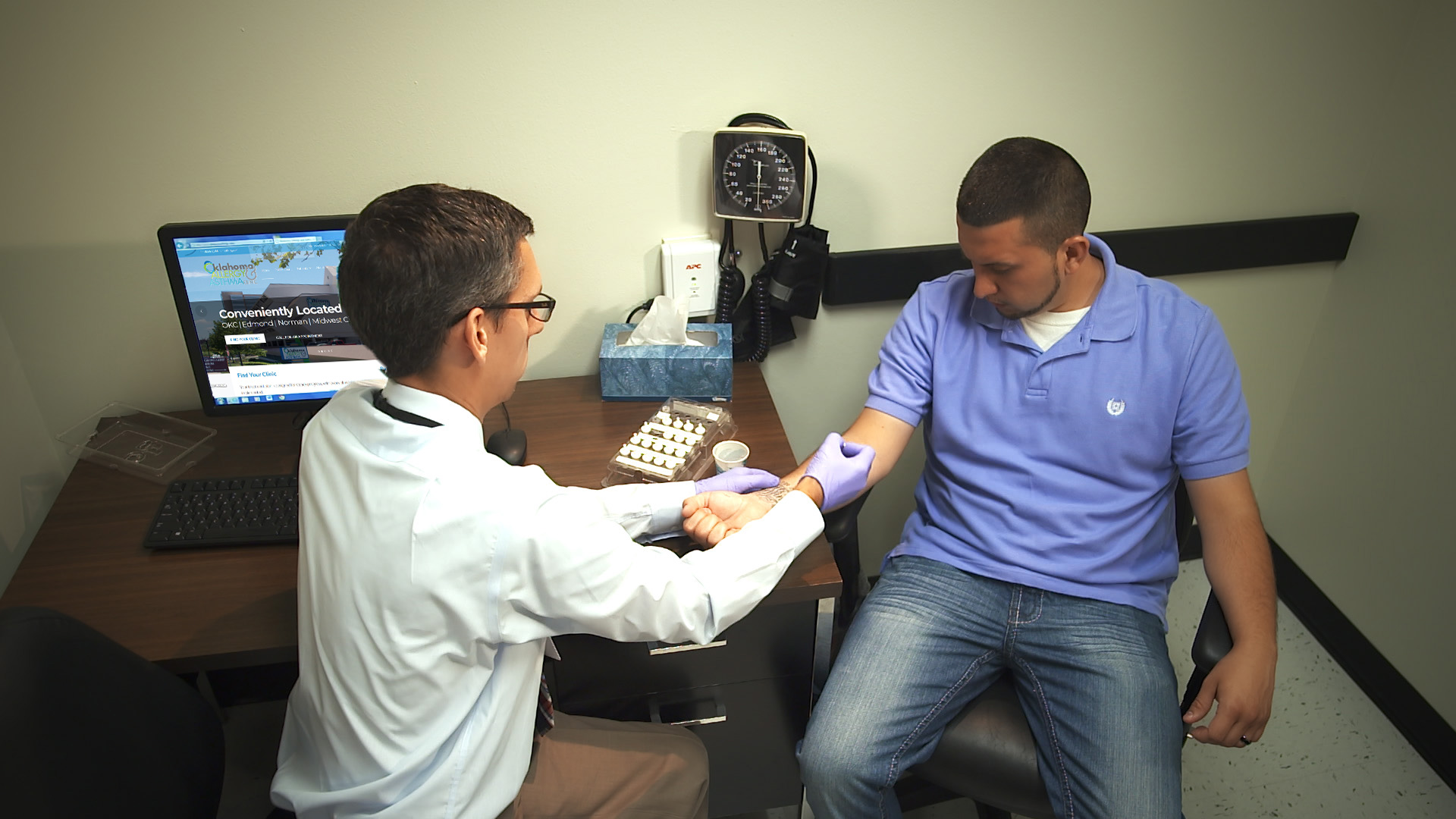 Advanced,Compassionate CareALLERGYASTHMAfor the wholefamily
Advanced,Compassionate CareALLERGYASTHMAfor the wholefamily -
 Our MissionThe Oklahoma Allergy & Asthma Clinic is committed to professional, compassionate, patient-centered care of the highest quality.
Our MissionThe Oklahoma Allergy & Asthma Clinic is committed to professional, compassionate, patient-centered care of the highest quality. -


 Award WinningVoted Top and Best Allergist 2024
Award WinningVoted Top and Best Allergist 2024
The Oklahoman & Journal Record Newspapers
Find Your Clinic
Your treatment plan is designed for steady progress, with every phase promptly implemented.
We have five locations to serve you.
Patient Portal
Your online door to your doctor’s office.
OAAC Telemedicine Instructions
Please follow these simple instructions for your appointments with OAAC providers.
As the largest practice in Central Oklahoma dedicated to the treatment of allergy and asthma, the Oklahoma Allergy & Asthma Clinic’s board-certified allergists can test what allergies you have and then put together an individualized treatment plan. Dedicated to the treatment of pollen allergies, food allergies, and asthma, we can help you live your best quality of life.
Conditions Treated
Our goal is to get you back to enjoying a healthy and active life, even when suffering from allergies. From diagnosis to treatment, we offer the following services:

Allergies
An allergic reaction typically triggers symptoms in the nose, lungs, throat, sinuses, ears, lining of the stomach or on the skin. For some people, allergies can also trigger symptoms of asthma. In the most serious cases, a life-threatening reaction called anaphylaxis can occur. To diagnose allergies, we conduct a physical examination, skin tests, and go over your medical history. Management includes various medications, and immunotherapy such as allergy shots help build immunity or tolerance to allergens is a very effective form of treatment.

Asthma
Asthma is a chronic disease involving the airways in the lungs. These airways, or bronchial tubes, allow air to come in and out of the lungs and are always inflamed. The muscles around the airways can also tighten leading to recurrent cough, shortness of breath or wheezing. We offer different diagnostic tests to evaluate asthma, including pulmonary function testing, methacholine challenge, nitric oxide level measurements, and chest x-rays. Treatment may include trigger avoidance, medications, immune or biologic therapies.

Food Allergies
Food allergies occur with the body has an abnormal immune response from exposure to a food. The most common food allergies are to eggs, milk, wheat, peanut, soy, tree nuts and seafood. Food allergies are often misdiagnosed, so your provider will obtain a detailed history and perform selected allergy tests to ensure appropriate diagnosis. Tests may include a skin test or blood allergy testing, depending on the severity of your allergy and what the allergist feels your body can handle. Treatment plans will vary by patient, and many of those patients are required to have an epinephrine auto-injector with them at all times.
When to See an Allergist
Some signs that indicate you should see an allergist include:
- Itchy, red, watery, swollen eyes
- Nasal congestion, drainage, sneezing
- Ear pressure or itching
- Sore, itchy, swollen mouth or throat
- Tingling or swelling in the mouth
- Hives
- Cough
- Tingling or swelling in the mouth
- Wheezing
Contact an Allergist Today
Since 1925, the Oklahoma Allergy & Asthma Clinic is committed to your well-being and the best quality of life. Contact us today at (405) 235-0040, whether you are suffering from different forms of allergies or asthma. We are happy to help you get your health back on track.
We are here to help you live your best quality of life
What are you waiting for? Don’t spend another day being miserable.
It’s time to TAKE control and find RELIEF!
Claire E. Atkinson
M.D.Dean A. Atkinson
M.D., FCAAILaura K. Chong
M.D., FACAAI, FAAAAIMatthew M. Farley
M.D.Maya N. Gharfeh
M.D.Karen L. Gregory
DNP, APRN, CNS, RRT, AE-C, FAARCRichard T. Hatch
M.D., FACAAAI, FAAP, AAAAIBret R. Haymore
M.D., FAAAAI, FACAAI, FACPGregory M. Metz
M.D., FAAAAI, FACAAI, AE-CPatricia I. Overhulser
M.D., FAAP, AAAAI, ACAAIMeredith A. Pruitt
PA-CStefanie E. Rollins
APRN, CNP, AE-CShahan A. Stutes
M.D.Elisa C. Thompson
APRN-CNP, AE-CLatest News
Very High Allergy Alert Issued for Tree Pollen
Very High Tree Pollen Alert Continues for Fifth Straight Day
Very High Allergy Alert for Tree Pollen Continues
Oak Tree Pollen Drives Thirteenth Very High Alert This Season
The Best in Oklahoma


OAAC has been honored as the Best of the Best from Oklahoma Magazine, Best Local Top Allergist by the Oklahoman’s Readers’ Choice Awards, Top Workplace in Oklahoma City by the Oklahoman newspaper, with the Torch Award from the Better Business Bureau for Business Ethics and named Top Allergists by the Journal Record business newspaper.
Why follow OAAC on Facebook, Twitter & Instagram?
In addition to posting pollen and mold counts each day, OAAC also uses social media to post announcements or educational allergy news updates. Follow us:
on Facebook at
www.facebook.com/oklahomaallergyasthmaclinic/
on Twitter @okallergyasthma
on Instagram @oklahomaallergyasthmaclinic
Patients / Employees

Patient Portal
Your online door to your doctor's office.

How to Join Our Team
Learn more about available positions at the OAAC.














